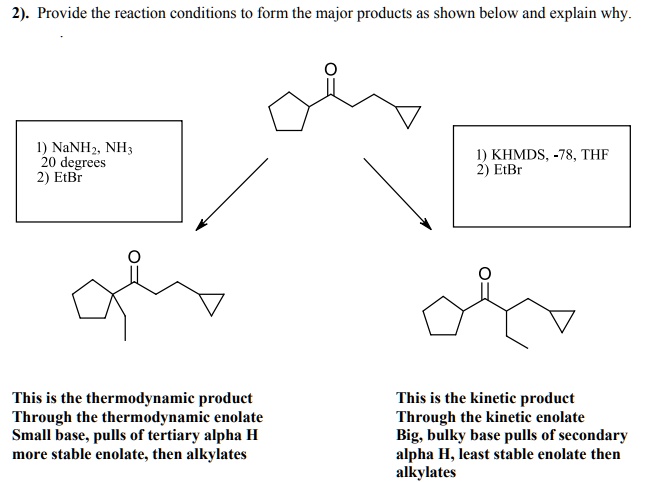
SOLVED: Provide the reaction conditions L0 form the major products as shown below and explain why NaNHz NH; 20 degrees EtBr 1) KHMDS, -78, THF 2) EtBr This is the thermodynamic product
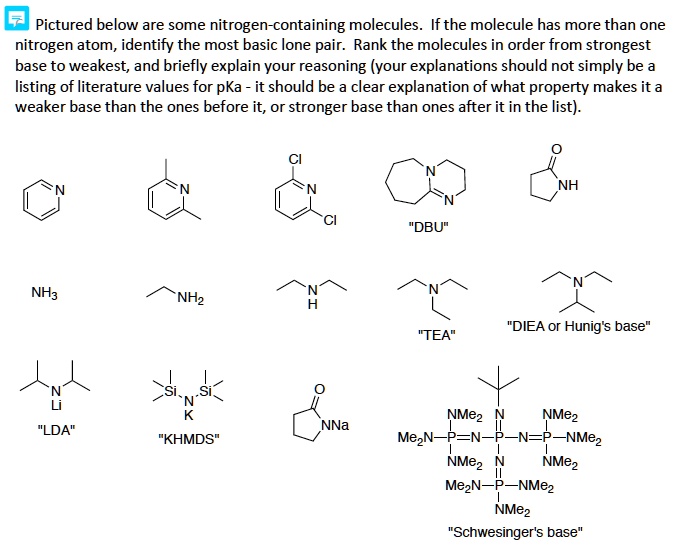
SOLVED: Pictured below are some nitrogen-containing molecules Ifthe molecule has more than one nitrogen tom, identify the most basic lone pair Rank the molecules in order from strongest base to weakest; and

Synthesis of π-Extended Fluoranthenes via a KHMDS-Promoted Anionic-Radical Reaction Cascade | Organic Letters
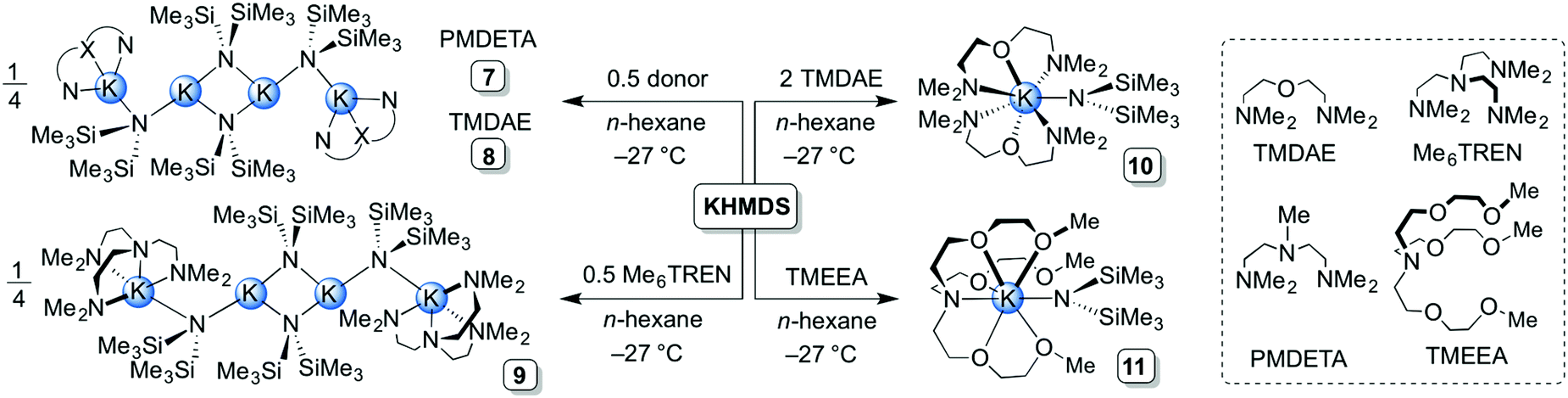
Exploring the solid state and solution structural chemistry of the utility amide potassium hexamethyldisilazide (KHMDS) - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT01118K

Potassium Amide‐Catalyzed Benzylic C−H Bond Addition of Alkylpyridines to Styrenes - Zhai - 2018 - Angewandte Chemie International Edition - Wiley Online Library

Pyridinylidenaminophosphines: Facile Access to Highly Electron‐Rich Phosphines - Rotering - 2020 - Chemistry – A European Journal - Wiley Online Library

Dimeric Potassium Amide-Catalyzed α-Alkylation of Benzyl Sulfides and 1,3-Dithianes | Organic Letters
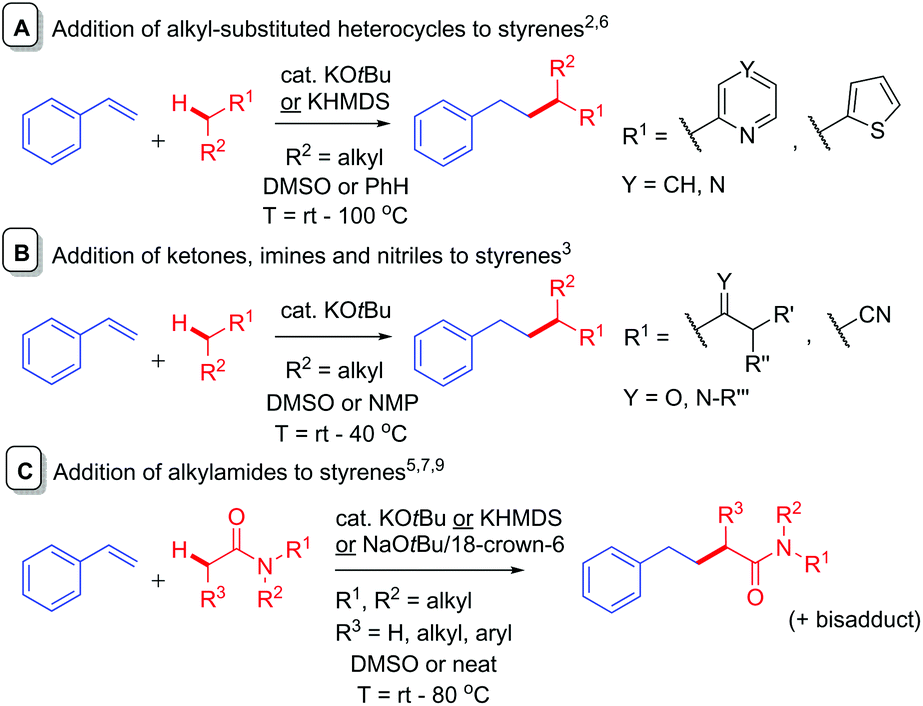
Base-catalyzed C-alkylation of potassium enolates with styrenes via a metal–ene reaction: a mechanistic study - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C9OB02495F

Potassium Base‐Promoted Diastereoselective Synthesis of 1,3‐Diols from Allylic Alcohols and Aldehydes through a Tandem Allylic‐Isomerization/Aldol–Tishchenko Reaction - Sai - 2021 - Chemistry – An Asian Journal - Wiley Online Library

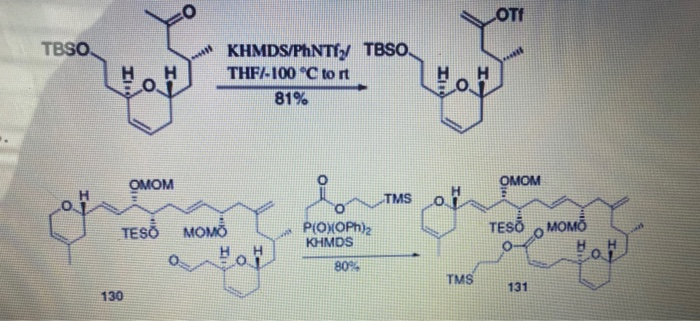






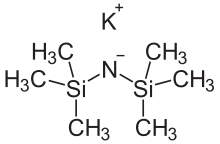


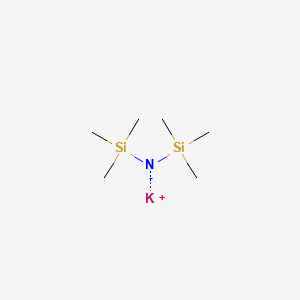

![a) Molecular structure of [(KHMDS) 2 (toluene)] ∞ 1, showing the... | Download Scientific Diagram a) Molecular structure of [(KHMDS) 2 (toluene)] ∞ 1, showing the... | Download Scientific Diagram](https://www.researchgate.net/publication/316670608/figure/fig2/AS:565319536447488@1511794088423/a-Molecular-structure-of-KHMDS-2-toluene-1-showing-the-contents-of-the.png)

